Blog
The challenges with electrochemical NO2 sensors in outdoor air monitoring

Article Details
Last Updated
14 September 2025
Published
23 July 2018
Category
Outdoor
Low-cost electrochemical nitrogen dioxide (NO2) sensors offer exciting new opportunities for fast and distributed outdoor air pollution measurements. The sensors are stable, long lasting, require very little power and are capable of measurement resolutions in the parts per billion (ppb) range. However, electrochemical NO2 sensors are susceptible to temperature (T), relative humidity (RH) and interfering gases, which can all impact the measurement.
With interest in smart cities and IoT devices skyrocketing over the past year, we have seen a sharp increase in customers wanting to purchase sensors that can be integrated into smart networks. A growing concern is that many integrators have limited understanding of how interferences impact electrochemical sensors and end users may be unaware of the potential false readings from these devices.
Here we will share some useful information on electrochemical sensors and how Aeroqual manages cross-interference for our GSE NO2 sensor.
A brief history
The oldest electrochemical gas sensors date back to the 1950’s and were originally developed for monitoring oxygen. By the mid 1980’s, miniaturized electrochemical sensors became available for the detection of many different toxic gases.
While electrochemical sensors have been the mainstay for workplace and hazardous gas leak detection for decades, their use in outdoor air monitoring presents different challenges. They need to accurately measure the analyte of interest at much lower concentrations, be selective towards the gas of interest and have the ability to show consistent readings over longer periods of time. The evolution of electrochemical sensors towards outdoor applications has come largely from advances in sensor technology and improvements in these key areas.
Nowadays, electrochemical gas sensors are available for just about any target gas, and they are used extensively in many stationary and portable monitoring applications around the world.
Construction and theory of operation
An electrochemical gas sensor measures the concentration of a target gas by using oxidation or reduction reactions to generate a positive or negative current flow through an external circuit. A typical electrochemical sensor consists of a “sensing” electrode, a “counter” electrode and in most cases a “reference” electrode. These components are combined inside the sensor housing with a liquid electrolyte. A commonly used electrolyte is sulfuric acid. Gas that comes into contact with the sensor passes through a hydrophobic membrane and diffusion limiting orifice and eventually reaches the surface of the “sensing” electrode. At the surface of the “sensing” electrode an oxidation or reduction reaction occurs, which results in a current between the “sensing” electrode and “counter” electrode. The magnitude of this current is proportional to the gas concentration.
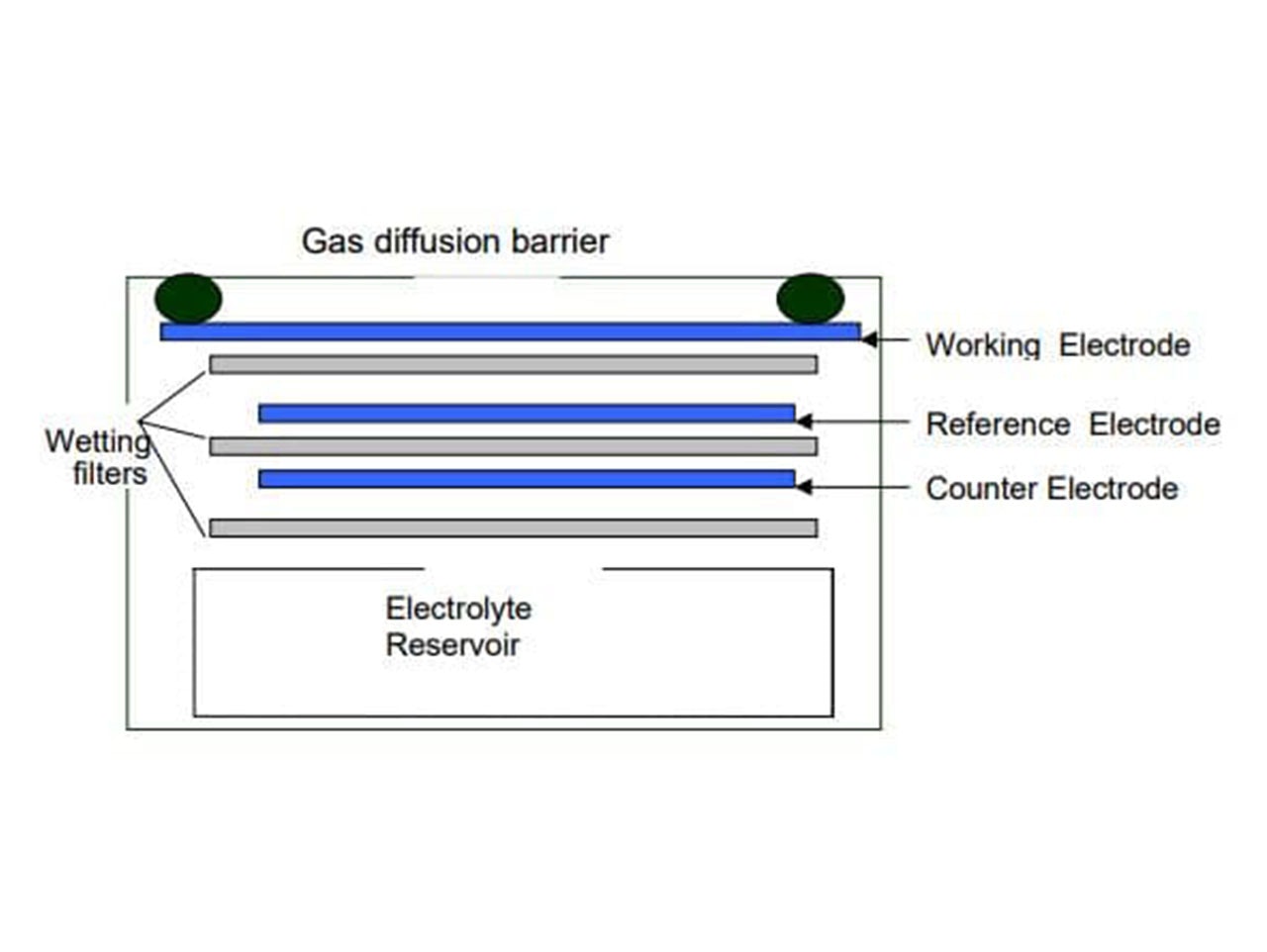
Typical Electrochemical Sensor Setup (photo credit: Alphasense, ‘How Electrochemical Gas Sensors Work’)
The construction of an electrochemical gas sensor involves careful selection of electrode materials, electrolyte composition and gas inlet design. While the appearance of sensors used to detect various gases may be similar, their internal structures are different and are the result of a compromise between various performance parameters. Consequently, one can expect varying performance from different sensor types and manufacturers in terms of sensitivity, selectivity, response time and operating life.
Environmental interference
Electrochemical sensors can be sensitive to interferences from changes in temperature (T), relative humidity (RH) and pressure (P), especially when operating at the lower limit of their sensing capabilities.
Temperature
Electrochemical sensors depend on chemical processes whose rates are proportional to temperature. Therefore, the output of most electrochemical sensors will vary with temperature. Some form of temperature compensation is recommended if the sensor is to provide reliable readings over a wide range of outdoor conditions.
Relative humidity
The electrolyte within the cell is separated by a hydrophobic membrane that prevents the solution leaking out of the sensor. However, water vapor in the air can pass through, just like other gas molecules can. This creates an issue in both high and low humidity. In high humidity conditions excess water vapor can build up and cause leakage. In low humidity conditions the sensor can dry out. These changes in the viscosity of the electrolyte affect the sensor output and subsequently the gas reading.
Pressure
Electrochemical sensors are minimally affected by pressure changes. It is important to keep the entire sensor within the same pressure since differential pressure within the sensor can cause sensor damage.
Interfering gases and cross-sensitivity
In a perfect world, an electrochemical sensor would only respond to the gas it is targeted to monitor. A nitrogen dioxide (NO2) sensor would only detect NO2 and a carbon monoxide (CO) sensor would only detect CO and so on. While electrochemical sensors are designed to be as specific as possible, most will respond in some manner to gasses other than the target gas. This is called “cross-sensitivity” and is a result of the interfering gas also reacting on the sensor electrode.
In some cases, the interfering effect is positive, and the user can get the impression that more of the target gas is present than actually is. In other cases, the interference is negative, and the user will see readings that are lower than the actual target gas concentration. This can result in negative numbers in the data set and the user may incorrectly elect to perform a zero calibration or assume there is a problem with the instrument.
In some situations when a gas that causes negative cross-sensitivity and the target gas exist together, you have a cancelling effect. This can be potentially dangerous if you are unaware of this phenomenon or are using equipment for high risk applications, like confined space monitoring.
Obviously, calibration of an electrochemical NO2 sensor using NO2 gas does not mean it will only give a NO2 response in air – the cross-sensitivity issue must be addressed first.

The electrochemical NO2 sensor problem: ozone cross-sensitivity
Most electrochemical NO2 sensors are extremely (almost 100%) cross-sensitive to the presence of oxidising gases like ozone (O3) and chlorine. Chlorine is not commonly present in ambient air except near sources. Ozone and NO2 are nearly always both present in outdoor air, especially during summer when sunlight, VOC and nitric oxides combine to form O3. It is important to be able to get exact concentration readings of O3 and NO2 separately for a couple of reasons:
- Source attribution. NO is generally related to traffic, which reacts with O3 to form NO2; while ground-level O3 forms when VOC and nitric oxides mix in the presence of sunlight.
- Different regulatory levels. Both O3 and NO2 are listed as criteria pollutants, as they pose a serious risk to human health.
Possible solutions to NO2 cross-sensitivity
There are two main ways to reduce the effect of cross-sensitivity on electrochemical NO2 sensors: the use of selective filters (chemical scrubbing) or correcting for the O3 interference (e.g. using Aeroqual’s selective GSS O3 sensor alongside the GSE NO2 sensor).
Selective filters (chemical scrubbing)
Some NO2 sensors include an internal filter designed to remove O3 and other oxidizing gas interferents before they reach the sensing electrode. Thus, the sensor is not affected by the presence of O3. While inclusion of a filter (such as MnO2) is frequently able to increase specificity, the filters have a finite lifespan because they lose their effectiveness before the operating life of the sensor is reached. Furthermore, most chemical scrubbers also reduce sensor sensitivity.
Pros
- Increased specificity
Cons
- Scrubbers are not selective
- Scrubbers efficiency reduces over time
- Sensitivity can be reduced

Correction using a selective ozone sensor
This technique applies a selective O3 sensor (such as Aeroqual’s GSS O3 sensor) to correct for the O3 interference on an electrochemical NO2 cell. The NO2 sensor measures a combination of NO2 and O3 (cross-interference effect), while the GSS is highly selective to O3. The weighted difference between the two sensors gives the NO2 concentration. Note that this approach only works if the O3 sensor is selective towards O3 and doesn’t respond to NO2.
Pros
- No scrubbing is needed (no consumables)
- No additional power requirement
Cons
- Only works if the O3 sensor is highly selective
Electrochemical NO2 sensor performance evaluation
Third party testing by recognized professional testing bodies, such as South Coast Air Quality Management District’s AQ-SPEC program, have found that many commercially available NO2 electrochemical sensors perform poorly when compared to federal reference method (FRM) equipment. Results from the AQ-SPEC testing are available here.
Aeroqual uses a GSE NO2 sensor and selective GSS O3 sensor in all its air quality monitoring stations, like the new AQY Micro Air Quality Station. The AQY has successfully applied the correction using a selective GSS O3 sensor approach in a number of field trials, achieving a very good correlation and high accuracy for NO2 (R2 >0.80 24-hr mean) against FRM analyzers. The secret to the AQY’s accuracy is the combination of an electrochemical NO2 sensor and our GSS metal oxide ozone sensor technology.
In summary
Electrochemical sensors are a proven technology that have been used for many years and will continue to be the cornerstone of gas detection sensors. The convenience of small and hearty sensors will always be of use for personal gas detection devices and outdoor air monitoring applications. When using them in your monitoring equipment, always consider the sensors’ limitations.
If you are interested to know more about Aeroqual products, or the services we offer, please get in touch. We’re always happy to hear from you.
Related products
Air monitoring made easy
Take the time and hassle out of your next project with a real-time air quality monitoring solution.
















